
On May 6, 2025, the new United States Government Policy for Dual Use Research of Concern and Pathogens with Enhanced Pandemic Potential (USG DURC-PEPP Policy) will go into effect.
The policy, which was first introduced in May 2024, requires life sciences research to be assessed at the point of grant proposal submission to ensure that any work meeting specific criteria has appropriate risk assessment and mitigation plans reviewed and approved by the federal sponsor PRIOR TO the PI receiving funding.
Please Note: This policy applies to all PIs proposing to work with or generate any replication-competent infectious agent, OR work with a toxin of any amount from the Federal Select Agents and Toxins List; it is NOT limited to only PIs who conduct research involving animals.
About the New DURC-PEPP Policy
The policy applies to all research – regardless of funding source – conducted under university auspices on the Ann Arbor, Dearborn, or Flint campuses that meets the criteria for DURC-PEPP research.
All Principal Investigators (PIs) proposing to work with or generate any replication-competent infectious agent, OR work with a toxin of ANY amount from the Federal Select Agents and Toxins List, must assess whether their project is “reasonably anticipated” to be within the Category 1 or Category 2 scope of research, as defined by the policy.
Category definitions are also outlined on the U-M Institutional Biosafety Committee’s (IBC) DURC-PEPP website and in the IBC DURC-PEPP Fast Facts Handout (available via PDF download using valid U-M login credentials). ![]()
![]() Links marked with a lock icon can only be accessed using valid U-M login credentials.
Links marked with a lock icon can only be accessed using valid U-M login credentials.
An initial review of U-M Institutional Biosafety Committee (IBC) applications that may be subject to the new policy has already begun. PIs who have been identified as part of this preliminary review should have received an email from the IBC requesting that they complete the DURC-PEPP Self-Assessment by Friday, April 4.
By the end of April, the IBC will reach out to all PIs with active IBC applications and request that they complete the same short self-assessment to determine whether additional steps may be necessary in order to comply with the new policy.
To streamline these processes moving forward, the U-M Proposal Approval Form (PAF) will be updated with a series of questions to ascertain whether the Self-Assessment is required. If so, the PI and Project Team will receive an automatic email notification with instructions on next steps for completing the assessment form.
Review the flowchart below for a more comprehensive overview (click to expand image)
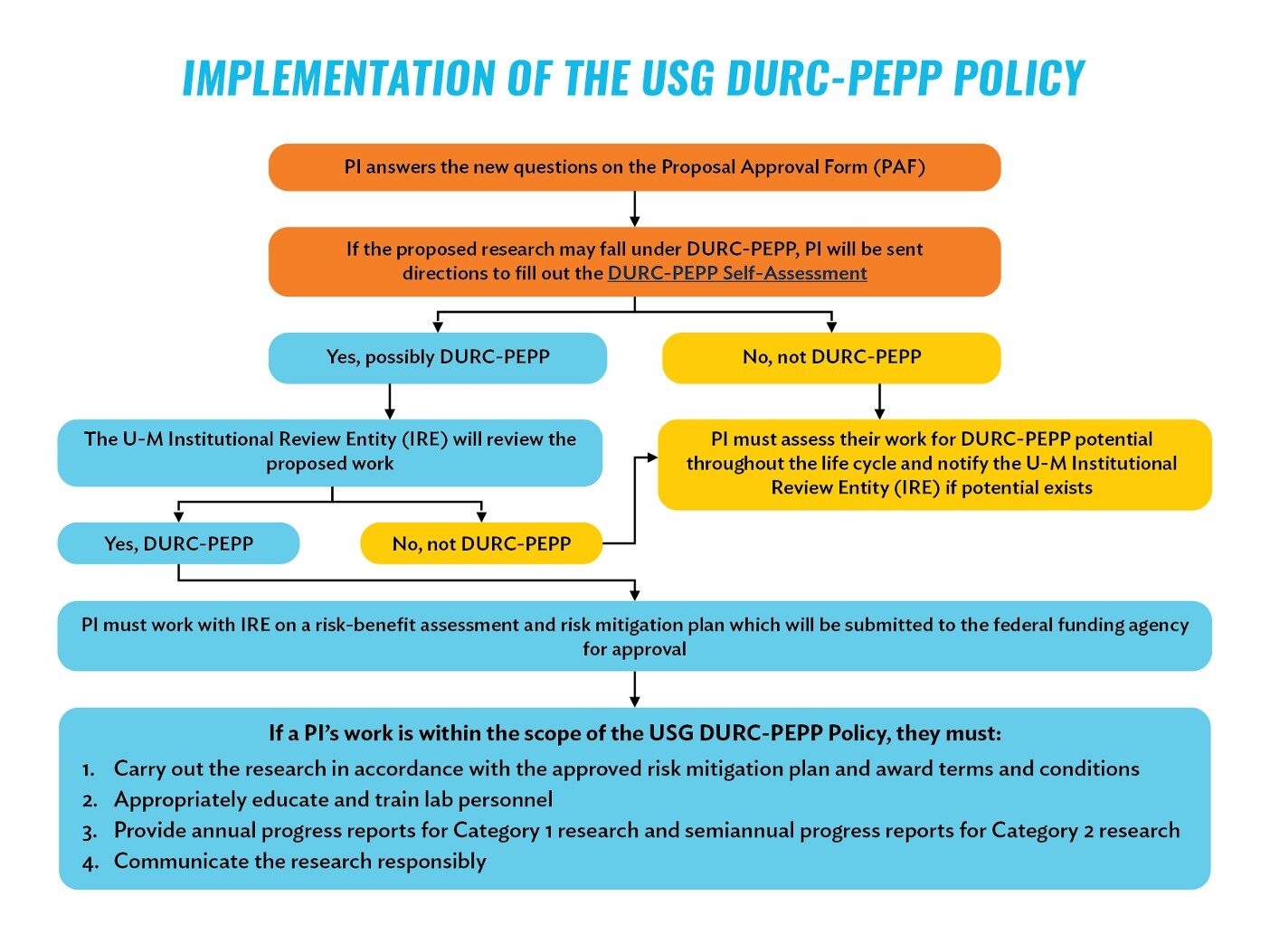
All PIs who conduct research on behalf of the university are responsible for knowing about – and remaining compliant with – all applicable institutional and U.S. government policies, requirements, and regulations pertaining to the use of biological agents and toxins in research.
As such, federal sponsors may delay the release of funds if a project identified as DURC-PEPP research does NOT fulfill all the requirements necessary for full policy compliance. This includes research that is funded or sponsored by federal grants, contracts, cooperative agreements, and other agreements.
Failure to follow the USG DURC-PEPP Policy may result in the suspension, limitation, or termination of federal funding, including the loss of future federal funding opportunities for the specific research being proposed as well as additional limitations on other life sciences research conducted at the institution.
Where to Learn More
Detailed information about this policy, including guidance for PIs and U-M’s implementation steps, can be found on the Institutional Biosafety Committee’s (IBC) DURC-PEPP Policy webpage. A list of frequently asked questions is also available via the IBC DURC-PEPP Fast Facts Handout (PDF download). ![]()
Questions about how or if your research may be impacted should be directed to Jacqueline Shields, Associate Director for Research Safety Compliance and the Institutional Contact for Dual Use Research, at [email protected] or (734) 936-3934.
![]() Links marked with a lock icon can only be accessed using valid U-M login credentials.
Links marked with a lock icon can only be accessed using valid U-M login credentials.