You are here
- Home
- Announcements
- Lessons Learned from Winter 2021 Semi-Annual Facility Inspections
Lessons Learned from Winter 2021 Semi-Annual Facility Inspections
 As the Fall 2021 semester quickly approaches and we welcome additional colleagues back into the lab, it is important to remember that every individual involved with animal care and use at the University of Michigan has an active role to play in ensuring animal welfare and maintaining programmatic compliance.
As the Fall 2021 semester quickly approaches and we welcome additional colleagues back into the lab, it is important to remember that every individual involved with animal care and use at the University of Michigan has an active role to play in ensuring animal welfare and maintaining programmatic compliance.
We firmly believe that increased transparency and information sharing will result in a more well-informed research community that is better prepared to assume collective responsibility for the humane treatment and judicious use of animals in research.
To help build a greater understanding of programmatic trends and key areas of focus for the upcoming term, we are sharing findings from the last round of semi-annual Institutional Animal Care & Use Committee (IACUC) facility inspections conducted in Winter 2021.
These inspections are one of the most important tools we have for evaluating our Animal Care & Use Program as well as offering areas for individual and collective improvement(s) in service to the animals under our care.
As such, please review and discuss these findings, suggestions for improvement, and additional resources with all members of your staff, including any students involved with research in your lab.
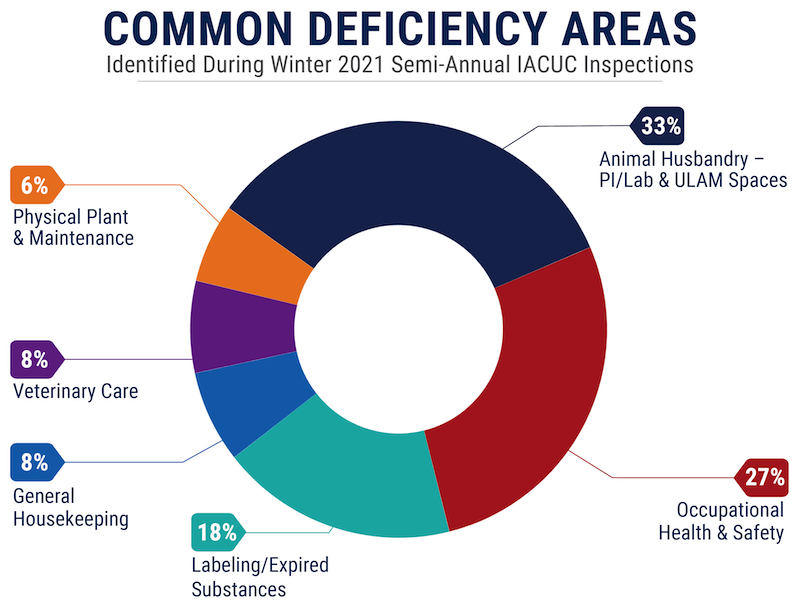
Common Deficiency Areas** Identified During Winter 2021 Inspections:
Examples:
- Sharpening/maintenance records for shears and guillotines not available
- Sanitation practices for all lab equipment that comes into direct contact with animals were not established and/or equipment was soiled
Suggestions for Improvement:
- Review the Policy on Maintenance of Guillotines, Scissors and Shears, which outlines requisite documentation for equipment servicing and maintenance
- Download the Quick Reference Sheet: Equipment Cleaning & Sanitization Guidance to familiarize yourself with the procedures and expectations for equipment sanitization and testing
Examples:
- Eyewash stations not flushed monthly
- Outdated or overfilled sharps containers
- Compressed gas cylinders not properly stored or maintained
Suggestions for Improvement:
- Review the Monthly Eyewash Inspection Checklist (pg. 73)
- Review EHS resources for sharps
- Download and review EHS Compressed Gas Guidelines and SOPs, which outline that storage of compressed gas cylinders requires sturdy chains or straps secured to a wall or cabinet, and/or use of a cylinder stand
Examples:
- Expired materials/drugs found
- Materials and substances not labeled
- Improper storage of controlled substances
Suggestions for Improvement:
- Review the Quick Reference Sheet: Managing Expired Drugs & Supplies for best practices on developing a system to monitor expired materials
- Create and maintain a regular schedule for reviewing substance expiration dates
- Ensure all materials/substances are labeled and dated, if necessary, upon arrival/order
- Remove expired substances or materials from circulation/use or appropriately label for non-recovery use only (as outlined by the Policy on the Use and Storage of Expired Drugs and Medical Materials in Animals)
Examples:
- Unsanitizable items (e.g., cloth chairs, unsealed wood) being used
- Labs found to be cluttered
Suggestions for Improvement:
- Fabric chairs in any areas where biological agents or other hazardous materials/chemicals are handled must be covered with a non-porous material that can be easily sanitized
- Review additional lab safety procedures in the EHS Chemical Hygiene Plan or consult with an EHS representative
- Select one or two lab members to regularly review, and be responsible for, housekeeping items/tasks
Examples:
- Post-operative monitoring sheets not available or incomplete
- Materials and substances not labeled
- Expired materials/drugs found
- Anesthesia machines not properly maintained
Suggestions for Improvement:
- Consult your IACUC protocol for details on expected post-operative/post-procedural monitoring. Contact your ULAM Faculty Veterinarian for additional information and/or training on post-operative care
- Download and print post-operative record templates
- Download the Quick Reference Sheet: Avoiding Common Mistakes When Completing Surgical Records for Mice and Rats
- Remove expired substances or materials from circulation/use or appropriately label for non-recovery use only (as outlined by the Policy on the Use and Storage of Expired Drugs and Medical Materials in Animals)
- Review the Policy on Maintaining Anesthetic Machines and Vaporizers, which states that anesthetic machines/vaporizers must be manually assessed at yearly intervals to ensure high-quality anesthesia for research animals and a safe working environment for personnel
- Utilize ULAM services for quarterly anesthetic machine servicing and certification
Examples:
- Humidity in animal rooms/housing locations inconsistent with acceptable ranges outlined in The Guide
Suggestions for Improvement:
- Work with your ULAM Husbandry Supervisor and/or Plant Operations staff to address any service or facility repairs in a timely manner
**Percentages above represent the number of overall deficient findings in a given area (e.g., Occupational Health & Safety – 27% denotes that,
of the total findings identified during Winter 2021 IACUC semi-annual facility inspections, 27% were related to occupational health and safety).
Additional Resources
In addition to the resources highlighted under each Suggestions for Improvement section above, you may also find the following tools helpful for assessing compliance and adherence to all policies, procedures, and guidelines within your lab:
| Resource | Description | |
|---|---|---|
| IACUC Self-Guided Evaluation of Animal Activities & Protocol Content Form |
An assessment form containing an easy-to-follow methodology for performing a self-guided evaluation of your protocol content, including the details of all animal activities | |
| IACUC Are You Inspection Ready? Checklist | A self-evaluation tool to help your lab plan for an IACUC inspection. Updated regularly to highlight key focus areas | |
| Aquatics Facilities – Are You Inspection Ready? Checklist |
A self-evaluation tool to help plan for an IACUC inspection of aquatics facilities | |
| What You Should Know About Reporting & Investigating Animal Welfare Concerns |
Includes information about the many ways suspected incidents of noncompliance can be reported, how allegations and reports are assessed within the program, details about the investigation process, and more | |
| Quick Reference Sheet: How to Run Training Reports in eRAM |
A high-level description of how PIs and Lab Contacts can easily monitor IACUC training requirements for their lab personnel | |
| Quick Reference Sheet: Guidelines for Rats & Mice Euthanasia Procedures |
A high-level guide for performing CO2 euthanasia procedures in adult mice. Note: these guidelines may not include all steps used in special circumstances. Always follow the procedures outlined in your IACUC approved protocol | |
| Archived AAALAC Chats | Not just for AAALAC preparation! These short 30-minute sessions provide a quick overview and resources on specific topics of interest to the U-M research community. Topics include controlled substances, rat and mice euthanasia procedures, equipment cleaning and sanitization, surgical records, daily husbandry and management of aquatics facilities, lab safety, protocol management, and tumor burden monitoring in rodents |
Don’t forget, we are here to serve as your partners and colleagues in conducting responsible research! Please reach out to us for guidance and assistance on how we can best support your research; we are here to help you and your team be successful.
| Unit/Individual | For assistance with... | |
|---|---|---|
|
Research Compliance Associates |
Processing protocols/amendments, policy guidance, assistance with understanding federal rules/regulations | |
| Quality Assurance Specialists (general inquiries may also be sent to ACU-QATeam@med.umich.edu) |
Inspection preparation, identifying/resolving protocol deviations, assistance with solving lab-specific problems | |
| ULAM Faculty Veterinarians (or email ulam-vets@umich.edu for questions that do NOT require immediate veterinary attention) |
Animal-related research techniques, treatment questions, general consults, protocol preparation | |
| ULAM Husbandry Managers (ulam-h-managers@umich.edu) |
Guidance and assistance with housing, facility, supply, and general procedure questions | |
|
Controlled Substances Monitors |
Use of controlled substances in research, including those projects involving animals | |
|
Institutional Biosafety Committee |
Research with potentially hazardous biologics, including but not limited to recombinant DNA and synthetic nucleic acid molecules |
|
|
Environment, Health & Safety |
Animal use and occupational health and safety |
Failure to appropriately follow policies and procedures, even those that may seem relatively small or insignificant, puts individual and animal safety at risk and could lead to serious citations for individual PIs, labs/lab staff, and the entire institution.
Maintaining a safe and secure environment for our peers and the animals entrusted to our care not only requires our constant attention and diligence, but it is also what is expected of us, both individually and collectively, when afforded the privilege of working with animals in research.
Questions?
Questions or concerns about semi-annual IACUC facility inspections, or any of the items outlined above, should be directed to the Animal Care & Use Office at acuoffice@umich.edu or (734) 763-8028.